Unit 3: Fourier Transform-Infrared Spectroscopy
Theory
Nearly all organic or inorganic compounds having covalent bonds absorb various frequencies of infrared radiation. Depending on the type of bonds present, a number of selected frequencies of IR radiation will be absorbed by a molecule.

With modern FT-IR instruments, it is relatively easy to record spectra from solids, liquids, gases and polymer films. In fact, a choice of sampling techniques exists for all states of matter. Some of the most common sample preparation methods are discussed in this section.
Techniques for Solids
(a) a solution in a suitable solvent such as CCl4,
(b) a suspension in liquid (mull),
(c) mixed and pressed into a alkali halide disc,
(d) reflectance techniques.
One of the easiest and the fastest methods to analyze a solid sample is by the mull technique. The solid sample is ground into a fine powder, suspended in a mineral oil such as Nujol, and the resulting mixture is ground to a smooth paste. Then, a small amount of paste is placed in between a pair of NaCl plates and the spectrum is recorded. The disadvantages to this technique are the lack of controlling the thickness of the paste in between the NaCl plates, and scattering of light by the solid particles. The latter can be minimized by using a liquid of the same refractive index as the solid sample.
The traditional technique of analyzing solid samples is by the Pellet Method. In this method, the sample is first ground into a fine powder. The powder is then mixed with dry KBr, and ground further. An agate mortar and pestle is normally used to grind the materials (agate is preferred since it is an inert material; KBr corrodes stainless steel). However, using an electrical mill for this purpose is much more convenient. A capsule made of polystyrene, stainless steel, or agate is used to contain the sample during the milling process. Stainless steel is popular, however, it should be noted that KBr will corrode this material. Small stainless steel balls are placed in the capsule with the sample and KBr and a stopper is used to close the capsule. The mill, depending on design, either vibrates the capsule, or rocks it back and forth at very high speed.
The finely ground material is then removed from the mill, placed in a KBr die and several tons of pressure is applied with a hydraulic press to coalesce the sample into a transparent or semi-transparent disk. Die sizes range from 1mm, for micro samples, up to the traditional 13mm diameter disks. When necessary, a vacuum line is connected to the die to remove entrained air, and to a limited extent, entrained moisture. As entrained moisture can cause the finished disk to be opaque the KBr powder should be kept dry by means of a desiccator or a heated oven. Moist samples are not suitable to this technique as they will cloud the disk. Such samples should be dried before analysis.
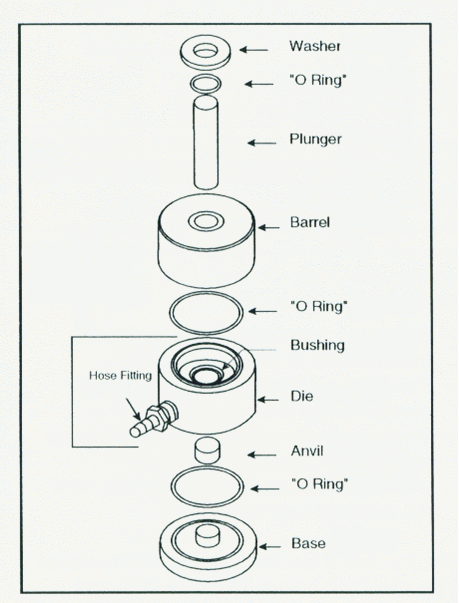 Assembly and Loading of Sample Step 1 • Place the base on a flat surface and insert the ‘0-ring in the groove • Place the die assembly over the post in the base. • Insert the anvil POLISHED SIDE UP into the bushing of the die. Step 2 • Load the prepared sample through the bushing hole and onto the anvil. • Level the sample matrix material using a microspatula or a clean, dry, glass rod. Step 3 • Place the second "0-ring" fl the groove and place the barrel on top. • Insert the plunger and place the "0-ring" and washer around the plunger. Pressing the Pellet • Place the complete die in a hydraulic press and connect the vacuum line. • After two minutes under vacuum, press the pellet. • After an additional two minutes, release the pressure and the vacuum • Wait one minute before removing the die from the press. Conversion for 13mm Die: lbs. total load = 0.205 x psiExamples: 25,000 psi - 5125 lbs. total load (gauge reading) Caution: DO NOT exceed 12,000 pounds total load on the gauge (60,000 psi at sample) |
Techniques for Liquids
solution in a suitable solvent such as CCl4,
a thin film of pure liquid between salt plates,
(c) reflectance techniques.
Usually for liquid samples as a solution, a demountable sodium chloride cell is used. The cell consists of two NaCl plates situated in between two supporting metal plates. At the two ends of the cell, there are two openings which are used to fill up the cell. Spectra of neat liquids can be recoded as a thin film pressed between two salt plates.
Techniques for gases
a gas cell with alkali halide windows,
a solution in a suitable solvent.
Techniques for polymers
(e) a solution in a suitable solvent such as CCl4,
(f) a thin film,
(g) a thin film evaporated on an alkali halide plate,
(h) a thin slice cut from larger sample,
(i) reflectance techniques.
The use of Horizontal Attenuated Total Reflectance (HATR) in the characterization of Polymer Films
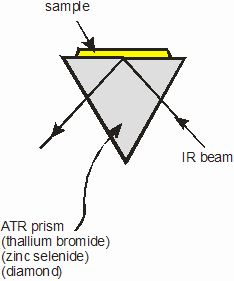 |
In the HATR system, a crystal of an IR transmitting material having a high refractive index, of 2.2 or more, is held in a horizontal plane. The IR beam from the spectrometer is directed into the crystal at an angle which exceeds the critical angle. Internal reflection takes place in the crystal. The sample is placed in optical contact with the surface at which this internal reflection occurs. At this reflection point, some energy is lost to the sample, corresponding to the absorption bands of the sample. The beam finally exits the crystal and mirrors of the accessory guide the IR the remaining energy to the instrument's detector. |
For samples of films and polymers, a flat plate system is used. The sample is placed on the crystal and a sample clamp used to provide optical contact. The sample clamp normally has a facility which enables it to accommodate samples of varying thickness and also to provide a range of contact pressures to optimize the experiment, by controlling band intensity.
Unit 3, Experiment 1: Characterization of organic compounds as thin films by FT-IR
Equipment

JASCO FT/IR-460 spectrometer
Reagents
* Polystyrene, polyethylene, nylon, saran, Teflon, polyester, mylar, and any other available thin films.
* Bring samples of wool, synthetic fabric such as polyester, soda bottles and cans.
Procedure
Record infrared spectra of at least five different thin films and two kinds of fiber by horizontal attenuated total reflectance (ATR) technique using JASCO FT/IR-460 spectrometer. [use polyethylene, polystyrene, nylon, saran, Teflon,
Cut one square inch pieces for soda bottles and cans, and fabric material.
Final report
Interpret spectra you recorded. Write the chemical structures of the polymers used. Assign vibrations to least three major peaks in each spectrum.
Record spectra from at least three samples you brought from home. Identify the polymers as best as you can.
Write a one page report on how infrared spectroscopy is used in forensic investigations.
|
Recycling Symbols |
|||||
| CODE | TYPE | NAME |
FORMULA |
DESCRIPTION | SOME EXAMPLES |
| PETE |
|
polyethylene terephthalate |
|
usually clear or green, sinks in water, rigid, glossy | soda bottles, peanut butter jars, vegetable oil bottles |
| HDPE |
|
high density polyethylene |
|
semi-rigid, sinks in water | milk and water jugs, juice and bleach bottles |
| PVC |
|
polyvinyl chloride |
|
semi-rigid, glossy, sinks in water | detergent / cleanser bottles, pipes |
| LDPE |
|
low density polyethylene |
|
flexible, not crinkly | 6-pack rings, bread bags, sandwich bags |
| PP |
|
polypropylene |
|
semi-rigid, low gloss | margarine tubs, straws, screw-on lids |
| PS |
|
polystyrene |
|
often brittle, glossy | styrofoam, packing peanuts, egg-cartons, foam cups |
Unit 3, Experiment 2: Characterization of organic compounds as solutions by FT-IR
The objective of this experiment is to record spectra from solutions, and then identify functional groups present in an unknown chemical provide by your TA.
Equipment
* Perkin Elmer Paragon 1000PC Fourier-transform infrared spectrometer.
* Demountable sodium chloride cell
Reagents
* Carbon tetrachloride, cyclohexane, ethyl acetate, methyl ethyl ketone, (10 mL of each)
* Unknown (labeled UNK? supplied by the laboratory instructor)
Procedure
1) Record infrared spectra of cyclohexane, ethyl acetate, and methyl ethyl ketone using a demountable NaCl cell. Use air (cardboard cutout) as background. If spectra of neat liquids are out of range, adjust their concentrations by diluting them with CCl4. (about 5 to 10 times dilution).
2) Obtain the infrared spectrum of the unknown.
3) Correlate functional groups to absorption peaks observed in all the spectra.
4) Determine the functional groups present in the unknown, and identify to which class of compounds it belongs to (e.g. alkanes, alkenes, etc.).
Report
Present your spectra of cyclohexane, ethyl acetate, and methyl ethyl ketone in an appropriate Figure. Correlate major abortion bands and to functional groups of the compounds tested.
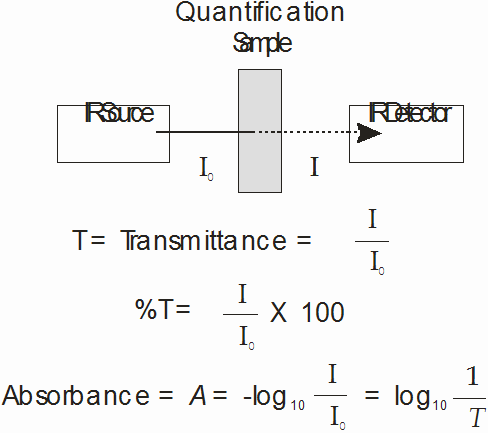
Unit 3, Experiment 3: Quantitative determination of Methyl Ethyl Ketone
The objective of this experiment is to determine the concentration of methyl ethyl ketone (MEK) in an unknown sample.
Theory: In the absence of chemical interactions, infrared absorbances should obey Beer’s Law, A = Elc, where A is the absorbance, E is the molar absorptivity, l is the path length, and c is the concentration. In this experiment you will prepare a standard curve by measuring the absorbance due to MEK in several standard solutions and plotting the data as absorbance vs. concentration. According to Beer’s Law, the plot should be linear. You will then record the spectrum of an unknown sample of MEK and utilize the standard curve to determine the unknown concentration.
Equipment
* Perkin-Elmer Paragon 1000PC Fourier-transform infrared spectrometer.
* Demountable sodium chloride cell
* 10 mL volumetric flasks (6)
* Syringe
Reagents
* Cyclohexane, methyl ethyl ketone, (10 mL of each)
* Solution of methyl ethyl ketone in cyclohexane of unknown concentration (labeled UNK-MEK? supplied by the laboratory instructor)
Procedure:
1) Prepare four standard solutions of MEK in cyclohexane of 0.3-1.5 % (v/v) range. Pure cyclohexane serves as the 0.0 % solution. Use the demountable liquid cell having a 0.2-mm gap between the NaC1 windows. The cell is filled using the glass syringe with a steel needle. The cell can be emptied using the plastic syringe to blow out any residual liquid. Clean the cell by flushing it with the organic solvent followed by the solution to be measured. Measure the most dilute solution first. Spent reagents, properly dispose to organic waste.
• Record a BACKGROUND scan using the empty demountable cell.
• Record a SCAN for each of the standards and the unknown. Save each spectrum.
• View the spectra in absorbance mode.
• Print a spectrum of the unknown only.
• Identify a peak whose size depends on the concentration of MEK.
• Overlay the spectra for the six standards and the unknown on one screen. Greatly expand the horizontal and vertical axes to obtain a view showing how the size of the peak of interest varies with concentration. Print this view.
• Select a frequency that is appropriate for quantification. Measure absorbance values at that frequency from the spectra of standards and that of the unknown.
• Use Excel to plot a calibration curve and calculate the concentration of MEK in the unknown solution.
Final report
• Determine the wavenumber of an absorption peak that is suitable for the quantitative analysis? On what basis did you select this peak? What functional group is responsible for this absorption peak?
• What relationship between absorbance and concentration is revealed from your data?
• What is the concentration of the unknown?
Unit 3, Experiment 4: IR Spectrum of benzoic acid
Equipment
* Perkin Elmer Paragon 1000PC Fourier-transform infrared spectrometer.
* KBr pellet apparatus (die, hydraulic press, grinder, etc.)
Reagents
* Potassium bromide
* Benzoic acid
* Nujol
Procedure
(1) The method of making a KBr pellet will be demonstrated by the laboratory instructor.
(2) Mix a few milligrams of benzoic acid with approximately 0.5 to 1 g of oven-dried KBr.
(3) Use a portion of this mixture to make the pellet, and obtain its spectrum.
(4) Grind approximately 0.1 g of benzoic acid into a fine powder.
(5) Add approximately 1 g of Nujol, and grind the mixture into a smooth paste.
(6) Place a small quantity of the paste in between two NaCl plates, and obtain its spectrum.
Final report
(1) Compare the spectrum of benzoic acid obtained by mull and KBr pellet techniques. Discuss the differences observed (if any).